NGOtiate er et dansk konsulenthus
Vi er en sjælden hybrid og en pioner inden for vores felt
Om os
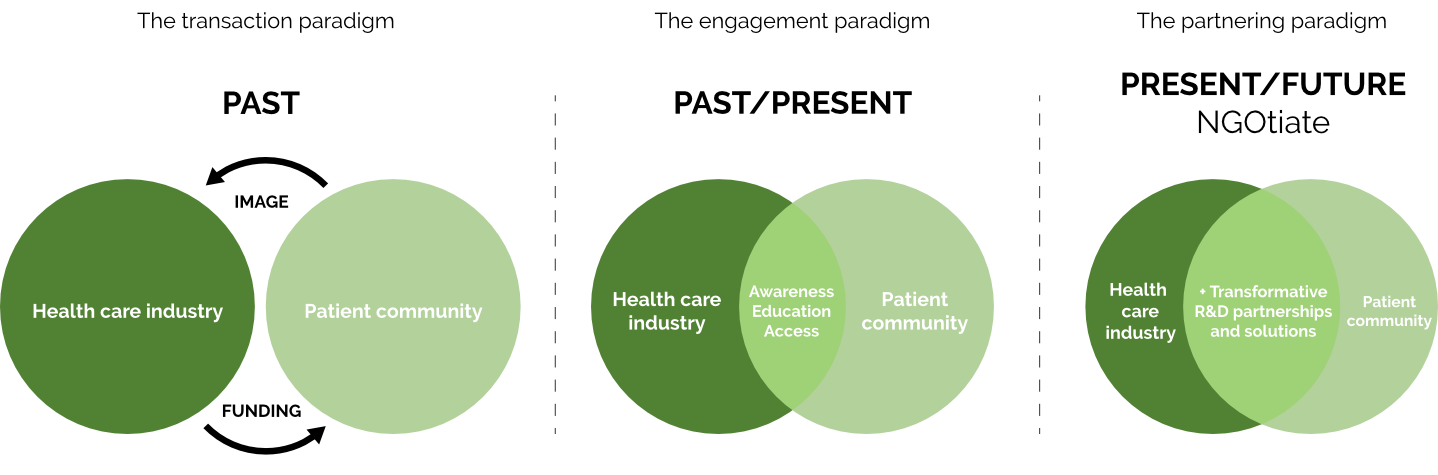
NGOtiate er et dansk konsulenthus grundlagt i 2010. Vi er en sjælden hybrid og en pioner inden for vores felt:
Vi specialiserer os i at hjælpe non-profit patientforeninger og farmaceutiske virksomheder verden over med at skabe større indflydelse gennem strategiske partnerskaber. Vi udvikler relaterede strategier, rammeværk og initiativer.
Vi skaber first-in-class og best-in-class innovationspartnerskaber og resultater for patienter. Vi forbinder de rette mennesker på det rette tidspunkt. Vi anvender data- og evidensbaserede metoder for at afdække uudnyttede potentialer og skabe ny viden.
Vi tænker og handler ud over ‘uopfyldte medicinske behov’ i vores mission om at spille vores rolle i at bane vej for løsninger, der imødekommer uopfyldte patientbehov.
Vi giver videre. Vores engagement går ud over vores forretning. Vi tilbyder gratis støtte til individer, der ønsker at etablere non-profit patient- eller familiemiljøer inden for underrepræsenterede sygdomsområder. Vi er her for at styrke de stemmer, der har brug for at blive hørt mest.
Vores ydelser
VÆR FORANDRINGEN
Vi hjælper patientforeninger og enkeltindivider med en vision!
Vi er her for at gøre din vision til virkelighed. Uanset om du drømmer om at opbygge et fællesskab fra bunden eller forstærke din gruppes/forenings indflydelse fx på forskning og udvikling eller adgang til nye og bedre behandlinger, står vi klar til at støtte dig hele vejen.
Fra Vision til handling
Drømmer du stort? Det gør vi også! Lad os hjælpe dig med at oversætte din idé til virkelighed – og til resultater, der gør en forskel for patienter og deres pårørende!
Skræddersyede kampagner
Uanset om dit mål er at skabe opmærksomhed, uddanne eller ændre status quo, hjælper vi med at designe effektive kampagner, som leverer på dit mål.
Markedsføring & Fundraising, der Virker
Har du brug for at udvide din rækkevidde eller sikre finansiering? Vi bygger en solid plan, og giver dig de nødvendige værktøjer til at få de ressourcer, du behøver for at blomstre.
Partnerskab i Innovation
Er du klar til at samarbejde om forskning og udvikling af medicin? Vi forbinder dig med de perfekte R&D-partnere (både farmaceutiske og non-farmaceutiske) for at skabe gennembrud sammen.
Din 24/7 Supportteam
Vi er altid her, når du har brug for os. Din mission er vores mission – sammen får vi det til at ske!
Vi hjælper farmaceutiske virksomheder i mål!
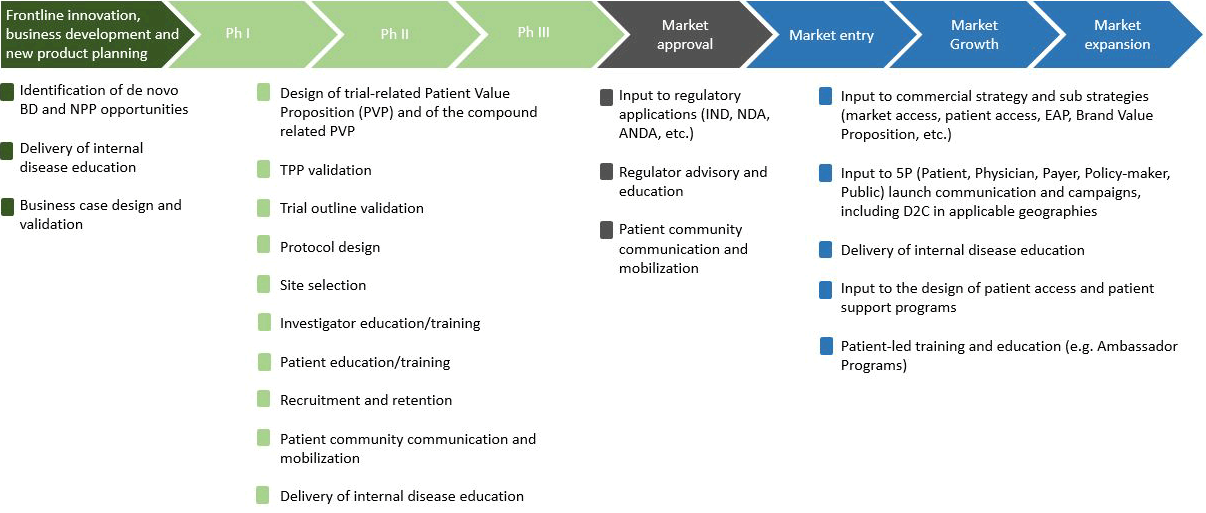
Strategisk Patient Enagement (SPE): Vi er her for at hjælpe din virksomhed med at opbygge, accelerere og mestre SPE. Fra strategi og rammeværk til patient partnerskaber der flytter nålen og driver resultater.
Opbygning af SPE-forståelse, kapabiliteter og rammeværk
Vi guider dig i at udvikle den operationelle infrastruktur og ekspertise, der er nødvendig for at levere effektiv SPE – fra grundlæggende principper til praktisk implementering og målbar succes.Skræddersyede SPE-strategier og metoder, der driver indflydelse og resultater
Uanset om du fokuserer på initiativer på tværs af hele virksomheden, porteføljemål eller program-specifikke mål, hjælper vi dig med at designe SPE-strategier og anvende validerede metoder, der fremmer succes på tværs af R&D, business development, regulatoriske indsendelser og produkt lanceringer.Forbinder dig med de rette patientpartnere
Vi hjælper dig med at finde og opstarte first-in-class og best-in-class samarbejder med de rette organisationer og enkeltindivider fra A-Å i din mission.Løbende, ekspertsupport – 24/7
Vi er altid her for at støtte dig, tilbyde ekspertvurdering og praktisk support.
Sådan er vi anderledes
partner paradigmET
Vores laserfokus er på det uudnyttede potentiale, der eksisterer i krydsfeltet mellem uopfyldte medicinske behov og uopfyldte patientbehov.
Vi er eksperter i at generere data og evidens, og i at omsætte relateret (ny) viden til drivkraft i innovation og i udviklingen af first-in-class og best-in-class løsninger for patienter.
Vi har over 20 års erfaring med at drive forandring i og for farmaceutiske virksomheder og patientorganisationer.
Vi er visionære do’ers. Vi er second-to-none partnere.
DEN DOKUMENTEREDE VÆRDI AF patient engagement
Optimere præstationen på tværs af værdikæden og i sidste ende slutbrugerprodukter (ref 1,6)
Positiv effekt (f.eks. økonomisk effekt) på rekruttering og fastholdelse i kliniske forsøg (ref 2,3,4)
Mulighed for forskere at lære fra patienter som eksperter i sygdomsoplevelse (ref 6)
Udfordre den etablerede tankegang – hvilket ofte fører til forbedret forskningsdesign, -levering og -formidling (ref 6)
Bredere virkninger af en ændret innovations-, forskningskultur og agenda (ref 5)
Kilder:
1: Wicks P, Richards T, Denegri S, Godlee F. Patients’ roles and rights in research.
2: Crocker Joanna C, Ricci-Cabello Ignacio, Parker Adwoa, Hirst Jennifer A, Chant Alan, Petit-Zeman Sophie et al. Impact of patient and public involvement on enrolment and retention in clinical trials: systematic review and meta-analysis
3: Boivin Antoine, Richards Tessa, Forsythe Laura, Grégoire Alexandre, Esperance Audrey, Abelson Julia et al. Evaluating patient and public involvement in research.
4: Levitan, Getz et al. 2018: Assessing the Financial Value of Patient Engagement 5 Staley, K. “Researchers don’t know what they’re missing”—the impact of patient involvement in research
6: Vat et. al (2019) Evaluating the “return on patient engagement initiatives” in medicines research and development
Kom i gang
step 1 - Ræk ud til os
Privatlivspolitik og Databehandlingsfraskrivelse
Denne Privatlivspolitik beskriver, hvordan vi indsamler, bruger og beskytter dine personoplysninger i overensstemmelse med den Generelle Databeskyttelsesforordning (GDPR) og andre gældende databeskyttelseslove. Ved at bruge vores tjenester, besøge vores hjemmeside eller indgå i en forretningsforbindelse med os, bekræfter du og accepterer de følgende betingelser:
2. Indsamling af Data
Vi indsamler personoplysninger, som du giver os direkte (f.eks. gennem formularer, kommunikation eller forespørgsler). Dette kan inkludere, men er ikke begrænset til:
- Navn
- E-mailadresse
- Telefonnummer
- IP-adresse
- Professionel information (f.eks. jobtitel, firmanavn)
3. Formål med Dataindsamling
Vi indsamler og behandler dine personoplysninger til følgende formål:
- At besvare dine forespørgsler og levere de ønskede tjenester.
- At sende dig markedsføringskommunikation (kun med dit samtykke).
- At opfylde vores kontraktmæssige forpligtelser og kommunikere om vores tjenester.
- At forbedre vores tjenester og sikre sikkerheden og effektiviteten af vores hjemmeside og forretningsdrift.
4. Retlig Grundlag for Dataindsamling
Vi behandler dine personoplysninger baseret på følgende retlige grundlag under GDPR:
- Samtykke: Når du giver os eksplicit samtykke til at behandle dine data.
- Kontraktmæssig nødvendighed: Når behandling er nødvendig for at opfylde en kontrakt eller aftale.
- Legitime interesser: Når behandling er nødvendig for vores legitime interesser (f.eks. forretningsdrift og markedsføring), medmindre dine rettigheder går foran disse interesser.
5. Deling af Data og Tredjeparter
Vi deler ikke dine personoplysninger med tredjeparter, undtagen i følgende tilfælde:
- Hvis det kræves ved lov eller for at beskytte vores juridiske rettigheder, kan vi videregive dine personoplysninger til myndigheder eller andre tredjeparter.
6. Opbevaring af Data
Vi opbevarer dine personoplysninger så længe det er nødvendigt for at opfylde de formål, der er beskrevet i denne Privatlivspolitik, eller som krævet ved lov.
7. Dine Rettigheder Under GDPR
Som den registrerede har du følgende rettigheder:
- Ret til adgang: Du kan anmode om adgang til de personoplysninger, vi opbevarer om dig.
- Ret til berigtigelse: Du kan anmode om at få rettet urigtige eller ufuldstændige data.
- Ret til sletning: Du kan anmode om at få dine data slettet under visse betingelser (f.eks. hvis data ikke længere er nødvendige).
- Ret til at begrænse behandling: Du kan anmode om at få behandlingen begrænset i visse tilfælde (f.eks. hvis nøjagtigheden af dine data er bestridt).
- Ret til indsigelse: Du kan gøre indsigelse mod behandlingen af dine personoplysninger til direkte markedsføring eller andre legitime grunde.
- Ret til dataportabilitet: Du kan anmode om at få dine data overført til en anden organisation i et struktureret, almindeligt anvendt og maskinlæsbart format.
- Ret til at trække samtykke tilbage: Du kan til enhver tid trække dit samtykke tilbage, hvis vi baserer behandlingen på samtykke.
Hvis du ønsker at udnytte nogen af disse rettigheder, bedes du kontakte os på [Indsæt kontakt-e-mail eller telefonnummer].
8. Datasikkerhed
Vi træffer passende tekniske og organisatoriske foranstaltninger for at sikre, at dine personoplysninger er beskyttet. Disse foranstaltninger er designet til at beskytte dine data mod uautoriseret adgang, ændring, videregivelse eller ødelæggelse.
9. Cookies
Vi bruger cookies for at forbedre din brugeroplevelse på vores hjemmeside. Ved at fortsætte med at bruge vores side giver du samtykke til brugen af cookies. Du kan administrere dine cookiepræferencer gennem dine browserindstillinger.
10. Ændringer af Denne Privatlivspolitik
Vi kan opdatere denne Privatlivspolitik fra tid til anden for at afspejle ændringer i vores praksis eller lovkrav. Eventuelle opdateringer vil blive offentliggjort på vores hjemmeside, og den opdaterede version vil være gældende, så snart den er gjort tilgængelig.
Ved at bruge vores tjenester bekræfter du, at du har læst og forstået denne GDPR-fraskrivelse og Privatlivspolitik. Hvis du har spørgsmål eller bekymringer om, hvordan vi behandler dine personoplysninger, er du velkommen til at kontakte os..